Precision-controlled melting process ensuring uniform alloy composition and superior metal purity

The melting of copper and copper-based alloys presents special problems. Molten copper dissolves both oxygen and hydrogen and on solidification, the oxygen and hydrogen can combine to form water vapor which causes porosity in the casting, Figs 16.1–16.4. Without the presence of oxygen, hydrogen alone may also cause gas porosity. Alloys containing aluminum form oxide skins which can cause problems in castings. In other alloys, traces of aluminum can cause defects and residual aluminum must be removed. Special melting and metal treatment techniques have been developed to deal with these effects. These include fluxing, degassing and de-oxidation treatments. Foseco supplies products for each of these treatments.
CUPREX formulations evolve oxygen to produce oxidizing conditions and a scavenging gas to remove most of the dissolved hydrogen, thus preventing the steam reaction which causes porosity in castings. CUPREX also forms a flux cover to prevent the pick-up of more hydrogen from the furnace atmosphere and remove non-metallic material.
CUPRIT neutral or reducing fluxes
The CUPRIT range is produced in briquette and powder form:
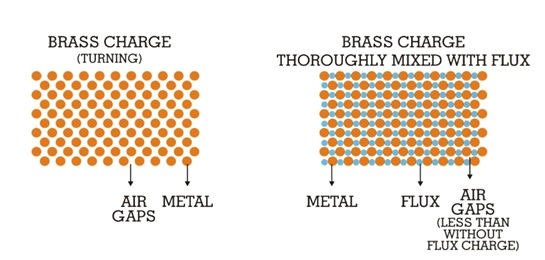
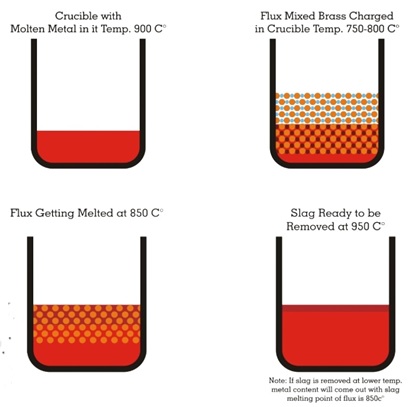
CUPRIT briquettes are pre-weighed while the powders are available in reweighed packets or in bulk. The main functions of CUPRIT are:
Rotary degassing of copper and its alloys:

LOGAS degassing units comprise a mixture of chemicals which, on contact with molten metal, decompose to release a steady stream of non-reactive gas. LOGAS is carefully dried and packed in foil, so the gas bubbles contain very little hydrogen and are able to flush out hydrogen from the melt.
De-oxidants for copper and its alloys
a) The ideal de-oxidant should function as follows:
b) It should combine with all the oxygen present to form a fluid slag.
c) De-oxidation products should not be entrained in the solidified casting.
d) Residual de-oxidant should not adversely affect the physical properties of the alloy and should prevent further oxidation during pouring.
Phosphorus satisfies most of these requirements, but a residual content of 0.025% is necessary to ensure adequate de-oxidation. This can seriously affect the conductivity of pure copper and causes embrittlement of high nickel bearing alloys.
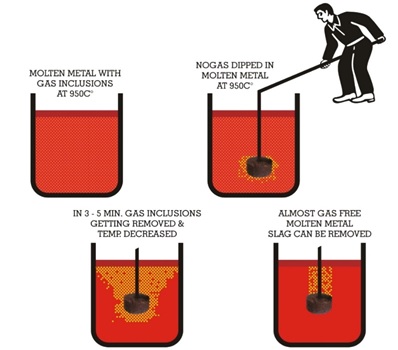
Hydrogen is removed from the melt by bubbling an inert gas through the melt. This can be done using argon or nitrogen using the Mobile Degassing Unit (see Chapter 6) or less effectively by injecting gas through a graphite tube immersed deep into the melt. 50–70 liters of gas are needed for each 100 kg of copper.
An alternative way to degas is to plunge LOGAS 50 briquettes into the melt. LOGAS is a granular material, strongly bonded and formed into a weighed unit with high surface area/volume ratio to ensure maximum contact area with the liquid metal. On contact with the metal, LOGAS 50 decomposes releasing a steady stream of non-reactive gas which flushes out the hydrogen. LOGAS 50 units are packed in foil, they are of annular shape having a central hole into which a refractory-coated steel plunger can be inserted,
Treatment takes from 3 to 10 minutes depending on the size of the melt. Some loss of temperature occurs during treatment, so the initial treatment temperature must be chosen accordingly. The minimum temperature practicable should be used.
A number of de-oxidants are available for copper. They combine with the dissolved oxygen in the metal forming stable oxides which float out of the melt. Phosphorus is the most widely used de-oxidant for copper and its alloys because of its effectiveness and low cost. It must be used sparingly with high conductivity copper since any residual phosphorus left in solution seriously lowers the conductivity of the copper.
The recommended practice is to use phosphorus to remove most of the dissolved oxygen and to complete the de-oxidation with a calcium boride or lithium-based de-oxidant.
The precise quantity of de-oxidant needed depends on the melting practice used. Simple tests can be made in the foundry to observe the solidification characteristics of the melt. Open-topped cylindrical test moulds having impressions about 75 mm high by 50 mm diameter are needed. They can be formed in a cold-setting resin or silicate sand mixture. When the melt is ready for de-oxidation, a sample of the copper should be ladled into one of the moulds and allowed to solidify. If the head rises appreciably as shown in Fig. 16.5a very gassy metal is indicated. DEOXIDISING TUBES DS containing phosphorus must be plunged and further test castings made. At the point when the quantity of phosphorus added results in a shallow sink in the head, it can be assumed that the residual phosphorus content of the melt is nil and a small amount, about 0.008% of oxygen, remains.
Effect of added elements
Aluminum: Unless added for a definite purpose, as in die-casting brass, it should be absent. It improves fluidity and definition, which is valuable in die castings, but it oxidizes readily causing oxide films and inclusions which may cause porosity and unsoundness in sand castings.
Iron: Small quantities have a grain-refining effect and increase hardness and tensile strength.
Lead: Improves machinability. Lead is insoluble in brass and exists as globules, which should be dispersed as uniformly as possible. It must not be allowed to segregate.
Manganese: Sometimes used as a de-oxidant, its effect is similar to iron.
Nickel: Improves the mechanical properties and increases corrosion resistance. It also has a tendency towards grain refinement.
Phosphorus: Combines with any iron present, increasing hardness. Reduces grain growth and improves fluidity.
Silicon: Makes founding more difficult but improves corrosion resistance and fluidity.
Tin: Raises tensile strength and hardness at the expense of ductility and improves corrosion resistance and fluidity.
ELIMINAL is a powdered flux range designed to reduce aluminum (and silicon) in copper-based alloys. Up to 0.5% Al may be removed from brass by means of ELIMINAL 2. Where higher levels exist, it is recommended that the charge is diluted with Al-free material to bring the Al content down to 0.5% or less. If the Al content is around 0.5%, the charge should be melted down under a cover of 0.5% by weight of ELIMINAL. This will also protect the zinc content of the melt. Before pouring, the metal should be brought to a temperature slightly higher than that required normally and 0.5% of ELIMINAL should be rabbled in or plunged to ensure maximum mixing, which is essential for efficient removal. The treatment is repeated until the Al content is reduced to the desired level. The metal is deoxidized immediately before pouring.
When aluminums have been removed to a low level, ELIMINAL will then remove silicon and manganese from copper alloys but at a slower rate than aluminums.
Nickel increases the solubility of hydrogen in copper melts, so it is necessary to melt under an oxidizing cover of CUPREX, followed by degassing with the Rotary Degassing Unit or LOGAS 50 to eliminate the hydrogen, then finally to deoxidize.
For a 100 kg melt, use 2 kg of CUPREX 1 blocks, degas with one LOGAS 50 briquette and deoxidize with DEOXIDISING TUBES MG (3 MG6 tubes for 100 kg). Note that Cu–Ni alloys may be embrittled by phosphorus, so DEOXIDISING TUBES DS should not be used.
Recommended pouring temperatures,